Standard Heat Unit Definition . The specific heat capacity is the amount of heat. Heat, as a form of energy, is quantified using several units that are essential in various scientific and. The heat capacity is the amount of heat, expressed usually in joules or calories, needed to change the system by 1 degree celsius. The standard heat of formation \(\left( \delta h^\text{o}_\text{f} \right)\) is the enthalpy change associated with the formation of one mole of a. What are units of heat? The standard unit of heat in the international system of units (si) is the joule (j). As all the energy is represented in joules (j), therefore, heat is also represented in joules. Another unit is the calorie (cal), with 1 calorie.
from blog.beamex.com
Another unit is the calorie (cal), with 1 calorie. As all the energy is represented in joules (j), therefore, heat is also represented in joules. The standard heat of formation \(\left( \delta h^\text{o}_\text{f} \right)\) is the enthalpy change associated with the formation of one mole of a. The standard unit of heat in the international system of units (si) is the joule (j). What are units of heat? The heat capacity is the amount of heat, expressed usually in joules or calories, needed to change the system by 1 degree celsius. The specific heat capacity is the amount of heat. Heat, as a form of energy, is quantified using several units that are essential in various scientific and.
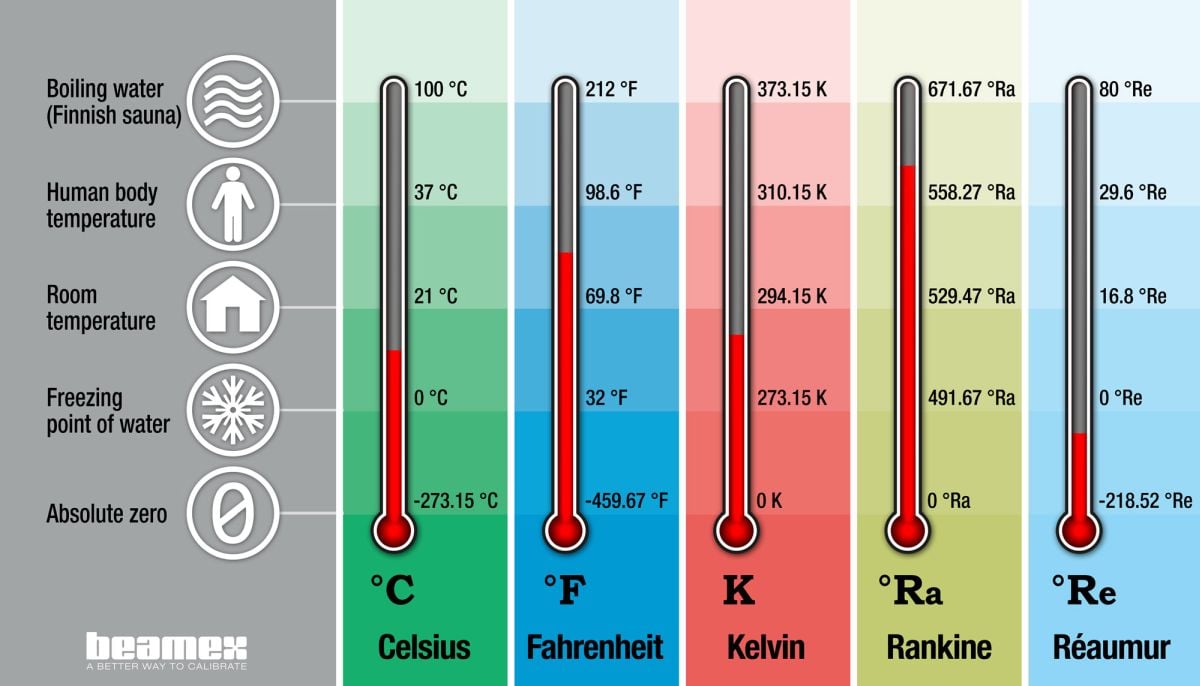
Temperature units and temperature unit conversion
Standard Heat Unit Definition Another unit is the calorie (cal), with 1 calorie. The heat capacity is the amount of heat, expressed usually in joules or calories, needed to change the system by 1 degree celsius. The standard heat of formation \(\left( \delta h^\text{o}_\text{f} \right)\) is the enthalpy change associated with the formation of one mole of a. What are units of heat? Another unit is the calorie (cal), with 1 calorie. Heat, as a form of energy, is quantified using several units that are essential in various scientific and. The specific heat capacity is the amount of heat. As all the energy is represented in joules (j), therefore, heat is also represented in joules. The standard unit of heat in the international system of units (si) is the joule (j).
From caliberfitness.com
HydraTherm, Standard Heating Unit Standard Heat Unit Definition The standard heat of formation \(\left( \delta h^\text{o}_\text{f} \right)\) is the enthalpy change associated with the formation of one mole of a. What are units of heat? The specific heat capacity is the amount of heat. The standard unit of heat in the international system of units (si) is the joule (j). Another unit is the calorie (cal), with 1. Standard Heat Unit Definition.
From www.tes.com
Specific heat capacity complete lesson (GCSE 19) Teaching Resources Standard Heat Unit Definition The heat capacity is the amount of heat, expressed usually in joules or calories, needed to change the system by 1 degree celsius. The standard heat of formation \(\left( \delta h^\text{o}_\text{f} \right)\) is the enthalpy change associated with the formation of one mole of a. The specific heat capacity is the amount of heat. Heat, as a form of energy,. Standard Heat Unit Definition.
From hvacsolvers.com
Heating Systems Explained Everything You Need to Know HVAC Solvers Standard Heat Unit Definition As all the energy is represented in joules (j), therefore, heat is also represented in joules. The heat capacity is the amount of heat, expressed usually in joules or calories, needed to change the system by 1 degree celsius. Heat, as a form of energy, is quantified using several units that are essential in various scientific and. The specific heat. Standard Heat Unit Definition.
From blog.beamex.com
Temperature units and temperature unit conversion Standard Heat Unit Definition Another unit is the calorie (cal), with 1 calorie. What are units of heat? The standard unit of heat in the international system of units (si) is the joule (j). The specific heat capacity is the amount of heat. Heat, as a form of energy, is quantified using several units that are essential in various scientific and. As all the. Standard Heat Unit Definition.
From www.numerade.com
SOLVED P5.16 Convection heat transfer data are often reported as a Standard Heat Unit Definition What are units of heat? The specific heat capacity is the amount of heat. As all the energy is represented in joules (j), therefore, heat is also represented in joules. Heat, as a form of energy, is quantified using several units that are essential in various scientific and. The standard heat of formation \(\left( \delta h^\text{o}_\text{f} \right)\) is the enthalpy. Standard Heat Unit Definition.
From alpineheat.com
American Standard Heating System Alpine Heating And Cooling Standard Heat Unit Definition Heat, as a form of energy, is quantified using several units that are essential in various scientific and. Another unit is the calorie (cal), with 1 calorie. The specific heat capacity is the amount of heat. The heat capacity is the amount of heat, expressed usually in joules or calories, needed to change the system by 1 degree celsius. The. Standard Heat Unit Definition.
From www.slideshare.net
Heat Capacity Standard Heat Unit Definition Another unit is the calorie (cal), with 1 calorie. What are units of heat? The standard unit of heat in the international system of units (si) is the joule (j). The standard heat of formation \(\left( \delta h^\text{o}_\text{f} \right)\) is the enthalpy change associated with the formation of one mole of a. As all the energy is represented in joules. Standard Heat Unit Definition.
From www.youtube.com
Standard Heat of Formation YouTube Standard Heat Unit Definition The heat capacity is the amount of heat, expressed usually in joules or calories, needed to change the system by 1 degree celsius. As all the energy is represented in joules (j), therefore, heat is also represented in joules. Another unit is the calorie (cal), with 1 calorie. Heat, as a form of energy, is quantified using several units that. Standard Heat Unit Definition.
From blog.beamex.com
Temperature units and temperature unit conversion Standard Heat Unit Definition The standard heat of formation \(\left( \delta h^\text{o}_\text{f} \right)\) is the enthalpy change associated with the formation of one mole of a. The specific heat capacity is the amount of heat. Heat, as a form of energy, is quantified using several units that are essential in various scientific and. The heat capacity is the amount of heat, expressed usually in. Standard Heat Unit Definition.
From huksefluxusa.com
What is a Heat Sink? Understand how they work. — HuksefluxUSA Standard Heat Unit Definition Another unit is the calorie (cal), with 1 calorie. The specific heat capacity is the amount of heat. As all the energy is represented in joules (j), therefore, heat is also represented in joules. What are units of heat? The standard heat of formation \(\left( \delta h^\text{o}_\text{f} \right)\) is the enthalpy change associated with the formation of one mole of. Standard Heat Unit Definition.
From www.slideserve.com
PPT Chapter 11 PowerPoint Presentation, free download ID831852 Standard Heat Unit Definition The standard unit of heat in the international system of units (si) is the joule (j). Another unit is the calorie (cal), with 1 calorie. The heat capacity is the amount of heat, expressed usually in joules or calories, needed to change the system by 1 degree celsius. What are units of heat? The standard heat of formation \(\left( \delta. Standard Heat Unit Definition.
From www.slideserve.com
PPT 18 Heat and the First Law of Thermodynamics PowerPoint Standard Heat Unit Definition As all the energy is represented in joules (j), therefore, heat is also represented in joules. The standard unit of heat in the international system of units (si) is the joule (j). The heat capacity is the amount of heat, expressed usually in joules or calories, needed to change the system by 1 degree celsius. Heat, as a form of. Standard Heat Unit Definition.
From www.numerade.com
SOLVEDDefine heat capacity and specific heat capacity. State their SI Standard Heat Unit Definition What are units of heat? Another unit is the calorie (cal), with 1 calorie. The standard unit of heat in the international system of units (si) is the joule (j). The specific heat capacity is the amount of heat. The standard heat of formation \(\left( \delta h^\text{o}_\text{f} \right)\) is the enthalpy change associated with the formation of one mole of. Standard Heat Unit Definition.
From sciencenotes.org
What Is Temperature? Definition in Science Standard Heat Unit Definition Heat, as a form of energy, is quantified using several units that are essential in various scientific and. The specific heat capacity is the amount of heat. Another unit is the calorie (cal), with 1 calorie. What are units of heat? The standard heat of formation \(\left( \delta h^\text{o}_\text{f} \right)\) is the enthalpy change associated with the formation of one. Standard Heat Unit Definition.
From www.youtube.com
Unit of heat energy SI unit of heat CGS unit of heat Joule Standard Heat Unit Definition What are units of heat? The heat capacity is the amount of heat, expressed usually in joules or calories, needed to change the system by 1 degree celsius. Heat, as a form of energy, is quantified using several units that are essential in various scientific and. The standard unit of heat in the international system of units (si) is the. Standard Heat Unit Definition.
From www.slideserve.com
PPT Thermal Physics PowerPoint Presentation, free download ID6948120 Standard Heat Unit Definition What are units of heat? Heat, as a form of energy, is quantified using several units that are essential in various scientific and. The standard unit of heat in the international system of units (si) is the joule (j). Another unit is the calorie (cal), with 1 calorie. The standard heat of formation \(\left( \delta h^\text{o}_\text{f} \right)\) is the enthalpy. Standard Heat Unit Definition.
From guidewiringpietism.z14..core.windows.net
Heating House With No Power Standard Heat Unit Definition The heat capacity is the amount of heat, expressed usually in joules or calories, needed to change the system by 1 degree celsius. The standard heat of formation \(\left( \delta h^\text{o}_\text{f} \right)\) is the enthalpy change associated with the formation of one mole of a. As all the energy is represented in joules (j), therefore, heat is also represented in. Standard Heat Unit Definition.
From wiringdbrae.z13..core.windows.net
American Standard Hvac Heat Pump Standard Heat Unit Definition The standard heat of formation \(\left( \delta h^\text{o}_\text{f} \right)\) is the enthalpy change associated with the formation of one mole of a. Another unit is the calorie (cal), with 1 calorie. What are units of heat? The specific heat capacity is the amount of heat. The heat capacity is the amount of heat, expressed usually in joules or calories, needed. Standard Heat Unit Definition.